|
Standard Free Energy, Enthalpy, and Entropy of Transfer of n-Bu4NBr from Water to Water-Acetone Mixtures at 298.15°K. 3) (c) the standard free energy of vaporization (d) the boiling point of 2-propanone. Calculate (a) the standard enthalpy of vaporization (b) the standard entropy of vaporization (use Eq. The vapor pressure of 2-propanone (CH COCH, more commonly known as acetone) is 67 Torr at 0.0☌ and 222 Torr at 25.0☌. 5, its standard entropy of vaporization at its boiling point is. To calculate the standard entropy of vaporization of acetone at its boiling point of 329.4 K (corresponding to 56.2☌), we note from Table 6.2 that its standard enthalpy of vaporization at its boiling point is 29.1 kjmol-1. The figure was constructed as follows first the standard enthalpy of transfer AH°t, obtained by Ahluwalia and co-workers (12) from pure water to Z2 = 0.30, was used in order to get the standard entropy of transfer function from the relation. Figure 4 and Table IV present the results obtained. 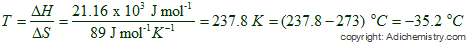
We shall discuss now the variation of the three main thermodynamic functions with solvent composition for the case of n-Bu4NBr- water-acetone system and shall extend this discussion to the n-Bu4NBr-water-THF system. g of acetone, CH3COCH3, condenses at its normal boiling point. 
(b) What is the entropy change of the surroundings when 10. The standard entropy of vaporization of acetone is approximately 85 J-K l-mol at its boiling point, (a) Estimate the standard enthalpy of vaporization of acetone at its normal boiling point of 56.2☌. What is the standard entropy of vaporization of acetone at its normal boiling point of 56.2☌.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
